Chinese blood pump to be tested in US

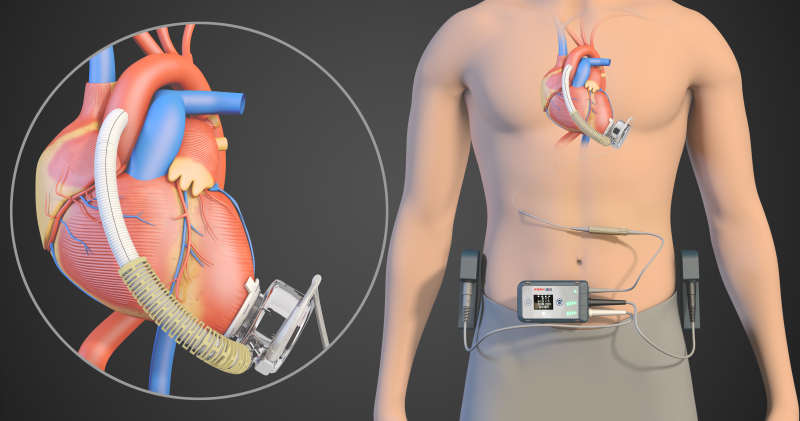
A blood pump that deploys magnetic levitation technology to better support heart function has been developed by a medical device company in Suzhou, Jiangsu province and will begin clinical trials in the United States next year, according to researchers.
The lemon-sized machine, also known as a left ventricular assist device, is implanted in a patient with a critical heart condition and assists in pumping blood.
It has been used in at least nine trial surgeries led by experts from Beijing's Fuwai Hospital, affiliated with the Chinese Academy of Medical Sciences.
He Xin, a patient who received the device in China, told China Central Television in June that he is now able to climb stairs and walk for hundreds of meters. In the past, he had to take long breaks after walking short distances.
The domestically developed device holds the potential to benefit even more patients with heart problems in China, cardiology experts said.
As the company pursues approvals for clinical trials overseas, Chen Chen, founder and CEO of CH Biomedical, which developed the pump and now manufactures it, said the device is proof of Chinese researchers' prowess in overcoming the complexity of making artificial hearts.
"Our foray into the foreign market of blood pumps is also likely to drive down costs of such devices and benefit a wider population who suffer from heart failure," Chen said.
At the core of the device is magnetic levitation technology, the same method that helps to reduce friction and significantly boosts the speed of trains.
"The technology essentially allows developers to reduce the size of the device, minimizing blood clots and bleeding, and thus improve overall efficiency and safety of the heart pump," Hu Shengshou, president of Fuwai Hospital, said at an academic conference in Beijing in November.
Global research into artificial hearts began in the 1960s, but "the fruition of many pioneers' efforts has only been seen over the last 10 years", said Wu Zhongjun, a professor of surgery at the University of Maryland's School of Medicine.
Wu said the new device, with its compact size and high compatibility with living tissue, will bring a wide range of concrete benefits to patients, including improving their quality of life, facilitating follow-up treatment and alleviating healthcare costs.
China was a latecomer to the development of artificial hearts, and some heart failure patients have died while awaiting donor hearts.
It is estimated that the number of people diagnosed with heart failure in China has climbed to 10 million in recent years, but only about 300 heart transplants are conducted annually.
"Chinese scientists did not delve deeply into this field until 2005," said Mo Ran, a surgeon at Nanjing Drum Tower Hospital in Jiangsu.
"The market potential of the device is significant for patients with severe heart failure because placing blood pumps inside their bodies is the best option to sustain their lives and maintain a good quality of life," Mo said.
He added that it is necessary to keep monitoring the health status of recipients and analyze their test results to fully validate the reliability of the device.
"Also, current trials are considered small scale," Mo said. "But the technology is certainly promising, and it's encouraging to see that China has finally caught up with global industry leaders in this area."




































