President Xi Jinping has emphasized the importance of taking into account the battlefield, the troops and the future in order to build world-class military medical universities, as the nation elevates the people's armed forces to worldclass standards.
 Pandas headed for Madrid in sign of bilateral friendship
Pandas headed for Madrid in sign of bilateral friendship
 Climate change blamed for heavy rain in the south
Climate change blamed for heavy rain in the south
 Reality show turns young artists into influential agri-preneurs
Reality show turns young artists into influential agri-preneurs
 Teacher-turned-livestreamer inspires viewers to fall in love with books
Teacher-turned-livestreamer inspires viewers to fall in love with books
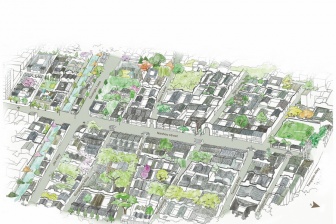 Bringing the history back to the homes
Bringing the history back to the homes

 京公网安备 11010502032503号
京公网安备 11010502032503号