'Nothing but cry'
A woman in her late 20s in Zhuhai, a city on the country's southern coast in
Guangdong, who called herself Xiao Cao, said she cried the whole night after
hearing of the ban.
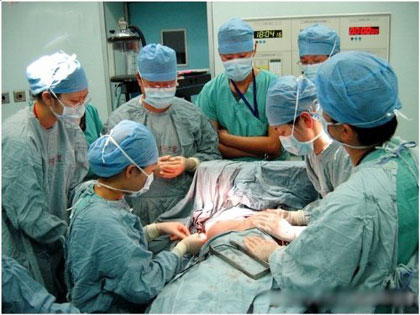
A woman goes under
knife to have her breasts enlarged.
[file] |
For countless nights after she had the liquid injected into her breasts 150
millilitres into each in 2003, she sat up for six or seven hours in her bed, not
able to sleep because her breasts ached so much. Blood seeped out of her breasts
after she gave birth in 2004.
And when she began lactating, she noticed it had a yellowish tinge.
"I was able to do nothing but cry because I was afraid that the injected
liquid was in my milk," she said.
The failure of the attempted beautification of her bodyline even gave rise to
quarrels between her and her husband. She began suffering from depression, and
the couple eventually divorced last year.
Xiao Cao has had several operations to try to expel the liquid from her
breasts, but they all failed. In the most recent one in July 2005 in Shenzhen,
she lay in the hospital bed for six hours and watched doctors and nurses unfold
the tops of her breasts and put their gloved hands into them and take out the
coagulating liquid.
"Doctors had to use a laser to scalpel the coagulations, which had grown into
the flesh," she said.
"I can never forget the smell of my flesh being burnt by the laser."
However, she still faces the threat of having to have her breasts removed
because only half amount of the liquid has been removed.
Big
profit
The Fu Hua Pharmaceutical Co Ltd was the Chinese distributor of a liquid for
similar use produced by a Ukrainian company, but co-operation between the two
sides broke up in 1998, according to Zhuo Xiaoqin, a lawyer and member of the
board of directors of the Chinese Health Law Society, who has been investigating
the Chinese company since 2000.
In 1999, the company asked for SFDA approval for the trial use of its new
product,
It maintained Ao Mei Ding was invented by its own researchers and a few
medical professors at Chinese universities.
It was approved for trial use in May 1999, trial production in December 1999
and then as a registered medical product in December 2000.